_____ take up most of the volume in atoms
Orbtitals take up most of the volume in atoms
Relative mass for an electron
1/1840
What letter represents the mass number?
A
What letter represents the atomic number?
Z
What type of ions have…
No. of electrons < No. of protons
Postive Ions
What type of ions have…
No. of electrons > No. of protons
Negative Ions
What holds the protons and neutrons?
Strong nuclear force
What holds electrons and protons together in atom?
Electrostatic forces of attraction
Why is the strong nuclear force stronger than electrostatic forces?
It overcomes repulsion between protons in nucleus
Strong nuclear force acts only over very ___ distances
SHORT distances (within nucleus)
What decides the chemical properties of an element?
No. & arrangement of electrons decides
Why do isotopes have the same chemical properties? (2)
- ∵ they have same electron configuration
- chemical properties depend on electrons
Isotopes have ___ _____ physical properties
slight different physical properties
Why do isotopes have slight different physical properties?
∵ physical properties depend on mass of atom
19th century: What did John Dalton say atoms were?
- Solid spheres
- Different spheres made different elements
- (All atoms of an element = same mass)
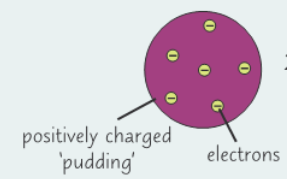
1897: What did J.J. Thomson discover and what did it show?
- Discovered the electron
- Showed atoms weren’t solid and indivisible
- (Model known as ‘plum pudding model’)
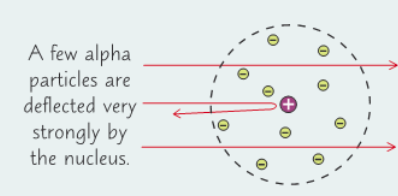
1909 - Ernest Rutherford: What did he find out?
Conducted the golden foil experiment:
- Fired positively charged alpha particles at a very thin sheet of gold
- Particles passed straight through gold & only small no. of particles were deflected backwards (pulm pudding model said = alpha particles would be deflected by the positive ‘pudding’ in atom)
- = developed into nuclear model of atom
- Tiny positive nucleus surrounded by ‘cloud’ of negative electrons - most of atom is empty space
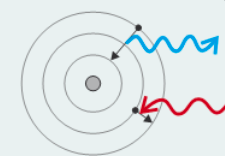
What was Niels Bohr’s model & discovery?
- Model: where electrons exist in shells or orbits of fixed energy
- Discovered: When electrons move between shells, electromagnetic radiation (with fixed energy or frequency) is emitted/absorbed
What have modern day scientists discovered & so what did they do?
- Electrons in same shell ≠ same energy
- Bohr model = wrong ∴ they refined it & added sub-shells
- (Isn’t perfect model but it’s simple and explains many experimental observations e.g. bonding & ionisation energy trends)
What are relative masses essentially?
Masses of atoms compared to carbon-12
Define Relative Atomic Mass (Ar) of an element (1x)
Average mass of an atom of an element on a scale where an atom of carbon-12 is 12
Define Relative Isotopic Mass
Mass of an atom of an isotope of an element on a scale where an atom of carbon-12 is 12
Define Relative Molecular Mass (Mr)
Average mass of a molecule on a scale where an atom of carbon-12 is 12
What does a mass spectrometer do and how?
- It determines the mass of separate atoms (or molecules)
- Works by forming ions from sample and then separating them according to the ratio of their charge to their mass

























