Cardiovascular Flashcards
(113 cards)
LO 1.1 Describe the factors influencing the exchange of substances between the blood in capillaries and the surrounding tissues
Area available for exchange - Determined by the Capillary Density (Capillaries per unit volume). Is highest in tissues that are most metabolically active. Note that not all capillaries are always perused.
Diffusion Resistance – The difficulty of movement through the barrier A major component of diffusion resistance is the distance over which diffusion must occur, the path length.
The Concentration gradient - Helps drive diffusion,This gradient does depend on the concentration of substances in the blood, but the more important variable is the flow of blood through the capillary. Unless blood is supplied at an appropriate rate, the gradients driving exchange will dissipate.
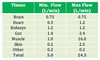
LO 1.3 List typical blood flows in ml/min/organ for major organs of the body, including the brain, kidneys, heart muscle, gut, skeletal muscle and skin, including both minimum levels and maximum levels.
LO 1.5 Describe the major functional components of the circulation
A Pump - The Heart Distribution
Vessels - The Arteries
Flow Control - Cardiac output must be distributed appropriately by restricting flow to those parts of the body that are easy to perfuse, so as to drive blood to parts that are not so easy to perfuse. Flow control is via resistance vessels, the arterioles and pre-capillary sphincters
Capacitance - Capacitance is the ability to cope with changes in the cardiac output. This is a store of blood that can be called upon to cope with temporary imbalances between the amount of blood returning to the heart and the amount that it is required to pump out. This store is the veins.
LO 1.6 What is the typical blood volume in the body and describe the distribution of blood volume over the major parts of the circulation.
At rest, the blood volume is about 5 Litres
11% - Arteries and Arterioles
5% - Capillaries
17% - Heart and Lungs
67% - Veins
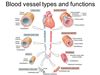
LO 1.7 Describe the different types of vessel, and be able to label the layers of each
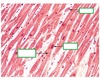
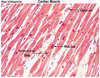
LO 2.2 Describe in general terms the properties of cardiac muscle that allow the heart to operate as a pump. How is it organised to best pump blood?
- Striations
- Branching
- Centrally positioned nuclei (1 or 2 per cell)
- Intercalated discs (for electrical and mechanical coupling with adjacent cells)
- Adherens-type junctions (to anchor cells and provide anchorage for actin)
- Gap junctions (for electrical coupling)
- In contrast with skeletal muscle, the T tubules of cardiac muscle are inline with the Z bands and not with the A-I band junction
LO 2.3 Define the terms Systole and Diastole, how long do each last?
Systole is the period when the myocardium is contracting, initiated by the SAN and lasting ~280ms.
Diastole is the relaxation in-between contractions, lasting about 700ms.
LO 2.4 What is the Spread of Excitation in Systole
- The SA node fires an action potential, which spreads over the atria causing atrial systole. The AP reaches the AV node, where it is delayed for about 120ms.
- From the AV node, excitation spreads down the septum between the ventricles
- Excitation spreads from inner (endocardial) to outer (epicardial) surface
- Ventricle contracts from the apex up, forcing blood towards the outflow valves.
LO 2.5 Describe the main differences between the right and left heart
The left side of the heart has a thicker myocardium, as it must generate the force to get blood around the entire body compared to the right side that must only get it around the lungs.
The left heart has the mitral valve whereas the right has the tricuspid valve.
The right side of the heart has the pacemaker, the SA node.
LO 2.6 Describe the sequence of pressure and volume changes in the atria and ventricles over a complete cardiac cycle in the normal individual. What is the way that ventricles contract in the heart?


LO 2.8 Explain the Origin of the 1st and 2nd Heart Sounds (also 3rd and 4th)
First Sound - As the AV valves closes, oscillations are induced in a variety of structures, producing a mixed sound with a crescendo-descendo quality – ‘lup’
Second Sound - As the semi-lunar valves close oscillations are induced in other structures, including the column of blood in the arteries. This produces the sound of shorter duration, higher frequency and lower intensity than the first – ‘dup’
A 3rd sound may be heard early in diastole, associated with heart failure or athletes.
A 4th is sometimes associated with atrial contraction. In exercise, turbulent flow generates ‘murmurs’ in normal individuals, but at rest murmurs are associated with disturbed flow, e.g. through a narrowed valve or back flow through an incompetent valve.
LO 3.1 Describe the frequency and most common types of congenital malformation of the heart and great vessels
Congenital heart defects are common, with an incidence of 6-8 per 1,000 births.
The most common heart defects are Ventricular Septal Defects (VSD), followed by Atrial Septal Defects (ASD).
LO 3.2 Discuss Atrial Septal Defects
An ASD is an opening in the septum between the two atria, which persists following birth. They have an incidence of 67 in 100,000 live births. The foramen ovale exists to prenatally permit right -> left shunting of oxygenated blood and is designed to close promptly after birth.Failure of it to close, allows blood to continue to flow between the two atria postnatally. Because left atrial pressure > right atrial pressure, flow will be mainly from left -> right, meaning no mixing of deoxygenated blood with the oxygenated blood being pumped around the circulation. ASDs can occur almost anywhere along the septum, but the most common site is the foramen ovale (Ostium secundum ASD). An ostium primum ASD occurs at the inferior part of the septum, and is less common.
LO 3.2 Discuss Patent Foramen Ovale
PFOs are not a true ASD. PFOs may be present in ~20% of the population and are generally clinically silent, since the higher left atrial pressure causes functional closure of the flap valve. A PFO may however be the route by which a venous embolism reaches the systemic circulation if pressure on the right side of the heart increases even transiently. This is called a paradoxical embolism.
LO 3.2 Discuss Ventricular Septal Defect
VSDs are an opening in the Interventricular Septum. This most commonly occurs in the membranous portion of the septum, but can occur at any point. Since left ventricular pressure is much > than right, blood will flow left to right.
LO 3.2 Discuss Patent Ductus Arteriosus
The Ductus Arteriosus is a vessel that exists in the foetus to shunt blood from the pulmonary artery to the aorta before the lungs are functioning. This vessel should close shortly after birth as the pressure in the pulmonary artery drops following perfusion of the lungs. Failure to close leads to a PDA. Blood flow through a PDA will be from the aorta to pulmonary artery after birth (High to Low pressure). A Mechanical Murmur is heart constantly throughout systole/diastole, as pressure in the aorta is always greater than in the pulmonary artery. Although left to right shunting of blood does not cause cyanosis it can be problematic later on if untreated, with the extent of the problems depending on the degree of shunting. Chronic left to right shunting can lead to vascular remodelling of the pulmonary circulation and an increase in pulmonary resistance. If the resistance of the pulmonary circulation increases beyond that of the systemic circulation the shunt with reverse direction as pressures on the right side of the heart increase (Eisenmenger Syndrome).
LO 3.2 Discuss Coarctation of the Aorta
Coarctation of the Aorta is a narrowing of the aortic lumen in the region of the ligamemtum arteriosum (former ductus arteriosus). The narrowing of the aorta increases the afterload on the left ventricle and can lead to left ventricular hypertrophy. Because the vessels to the head and upper limbs usually emerge proximal to the Coarctation, the blood supply to these regions is not compromised. However blood flow to the rest of the body is reduced. The extent of the symptoms depends on the severity of the Coarctation. In very severe cases, an infant may present with symptoms of heart failure shortly after birth. In mild cases, the defect may be detected in adult life. Femoral pulses will be weak and delayed, with upper body hypertension.
LO 3.3 Discuss Tetralogy of Fallot
The Tetralogy of Fallot is a group of 4 lesions occurring together as a result of a single developmental defect placing the outflow portion of the interventricular septum too far in the anterior and cephalad directions. The four abnormalities are: - VSD - Overriding Aorta - Pulmonary Stenosis (variable degree) - Right Ventricular Hypertrophy (variable degree) Pulmonary stenosis causes persistence of the foetal right ventricular hypertrophy, as the right ventricle must operate at a higher pressure to pump blood through the pulmonary artery. The increased pressure on the right side of the heart, along with the VSD and overriding aorta allow right to left shunting and therefore the mix of deoxygenated blood with the oxygenated blood going to the systemic circulation, resulting in cyanosis. The magnitude of the shunt and level of severity depends on the severity of the pulmonary stenosis. Affected individuals may present with cyanosis in infancy, but mild cases can present in adulthood.
LO 3.3 Discuss Tricuspid Atresia
Tricuspid Atresia is the lack of development of the tricuspid valve. This leaves no inlet to the right ventricle. There must be a complete Right to Left shunt of all blood returning to the right atrium (ASD or PFO) and a VSD or PDA to allow blood to flow to the lungs.
LO 3.3 Discuss Transposition of the Great Arteries
Results in two unconnected parallel circulations instead of two in series. In this defect, the right ventricle is connected to the aorta and the left ventricle to the pulmonary trunk. This condition is not compatible with life after birth, unless a shunt exists to allow the two circulations to communicate. A shunt must be maintained or created immediately following birth to sustain life until surgical correction can be made. The ductus arteriosus can be maintained patent and/or an atrial septal defect formed.
LO 3.3 Discuss Hypoplastic Left Heart
In some cases the left ventricle and ascending aorta fail to develop properly resulting in a condition called Hypoplastic left heart. A PFO or ASD are also present and blood supply to the systemic circulation is via a PDA. Without surgical correction this is lethal.
LO 4.1 Understand the Anatomical and Pharmacological divisions of the autonomic nervous system (ANS)
The ANS is comprised of a series of 2 neurones – one pre-ganglionic and one post-ganglionic. The ganglion is the collection of cell bodies in the PNS . 1 cell has its cell body in the CNS. 1 cell has its cell body in the PNS.
Sympathetic Nervous System
Thoraco-Lumbar Outflow Nerve fibres have cell bodies in all 12 thoracic sections and the first 2 lumbar sections. Short pre-ganglionic nerve fibre – Long post-ganglionic nerve fibre May synapse at same level as origin (paravertebral origin), may synapse at different level to origin or may not synapse in paravertebral chain Pre-ganglionic neurones are cholinergenic (Ach) (Nicotinic) Post-ganglionic neurones express nicotinic receptors
Post-ganglionic neurones are noradrenergic (NA) (Two classes of adrenoceptors A(1 & 2) and B(1 & 2) Exception some synapses are cholinergic: perspiration and ejaculation pathways
Parasympathetic Nervous
System Cranio-Sacral Outflow Long pre-ganglionic nerve fibre – Short post-ganglionic nerve fibre Pre-ganglionic neurones are cholinergic (Nicotinic) Post-ganglionic neurones express nicotinic receptors Post-ganglionic neurones are cholinergic (Muscarinic, G-Protein coupled)
LO 4. 3 State the action of the sympathetic nervous system on blood vessels in different organs
The sympathetic branch of the autonomic nervous system innervates the smooth muscle in the walls of arteries, arterioles and veins. Except in specialised vessels (e.g. erectile tissue with parasympathetic innervation), sympathetic activity causes constriction of arterioles, vasoconstriction, via 1 adrenoceptors. There is constant activity of the sympathetic nervous symptom – Vasomotor Tone. This tone varies from organ to organ. In skin for example, vasomotor tone is high, so arterioles, pre-capillary sphincters and arterio-venous anastomoses are generally shut down. Variation in sympathetic outflow produces large changes in skin blood for, for thermoregulation. In skeletal muscles, vasomotor tone is high at rest, but in exercise is antagonised by vasodilator metabolites. In the gut, vasomotor activity is high until a meal is consumed, when various vasodilators produced in the gut tissue antagonise it. Circulation to the brain is virtually unaffected by sympathetic activity. The interplay between sympathetic vasoconstrictor tone and the action of vasodilator substances is the principal means by which the distribution of flow around the CVS is controlled. Vascular tone in the skin and skeletal muscle is also a mechanism for controlling the total peripheral resistance. Sympathetic outflow to blood vessels is controlled from the brainstem, via the ‘vasomotor’ centres in the medulla oblongata.
LO 4.4 State the action of the sympathetic and parasympathetic system upon heart rate and force of contraction
Both the parasympathetic and sympathetic branches of the ANS, which innervate both the SA and AV nodes, affect heart rate. Parasympathetic activity slows the heart rate and sympathetic speeds it up. Both are controlled by centres in the medulla oblongata, which themselves receive information from the Baroreceptors, which are located in the arch of the aorta and carotid sinuses. There is a basal activity of both systems at rest, but if all autonomic inputs are pharmacologically blocked, the intrinsic heart rate is ~100bpm. The normal resting HR of ~60bpm is produced because the parasympathetic dominates the sympathetic at rest. Initial increases in heart rate are brought about by reduction in parasympathetic outflow. Increasing Sympathetic outflow makes further increases. Sympathetic nerve fibres also innervate ventricular cardiac myocytes, increasing the force of contraction and cardiac output. Sympathetic – NA - B1 Receptors Parasympathetic – Ach – M2 Receptors Adrenaline from the adrenal medulla can also act on the heart