Amines Flashcards
Is an amide more reactive than a carboxylic acid? Do additional alkyl substituents make an amine more or less acidic? Master these topics and much more, and gain that confidence you need to ace any amine-related question on the MCAT. (30 cards)
Define:
amine
An amine is an organic molecule derived from ammonia (NH3) in which one or more hydrogen atoms has been replaced with a carbon chain.
To name an amine, remove the “-e” from the name of the parent alkane and replace it with “-amine.”
When is the prefix “amino-“ used to name an amine?
The prefix “amino-“ is used to name an -NH2 group as a substituent when the molecule contains higher-priority groups. For example, alcohols and carboxylic acids take priority over amines.
“Amino-“ is also used as a prefix when the -NH2 group is located on a non-terminal carbon.
What is the name of this molecule?

This molecule is butanamine.
To name an amine in which the NH2 group is on a terminal carbon, simply replace the “-e” of the parent alkane chain with “-amine.”
What is the name of this molecule?

This molecule is 2-aminopropanol.
The highest-priority group on this carbon chain is the -OH. Therefore, the molecule should be named as an alcohol and given the “-ol” ending. The “amino-“ prefix is used to denote the -NH2 group as a substituent on the chain.
When can an amine’s nitrogen atom be chiral?
A nitrogen can have chirality if it is bound to either three or four asymmetric groups.
Tertiary amines invert spontaneously at room temperature and exist as a racemic mixture. Quaternary amines do not invert and thus maintain their chirality.
Is the following molecule chiral, and will a solution of it at room temperature be optically active?
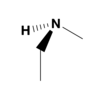
The molecule is chiral. However, it will not be optically active in solution.
This molecule is a tertiary amine bound to three different groups, so it is chiral. Tertiary amines spontaneously invert at room temperature, making a solution of this molecule racemic and not optically active.
What is the strongest type of intermolecular attraction that can be exhibited by an amine?
Amines can form hydrogen bonds.
Hydrogen bonding increases the boiling and melting points of amines relative to those of alkanes.
How do the boiling points of amines compare to those of alcohols?
Assume all compounds have similar molecular weight, chain length, and branching.
Amines tend to have lower boiling points than those of analogous alcohols.
This trend exists because alcohols exhibit stronger hydrogen bonding and thus have higher intermolecular forces.
How do the melting points of amines compare to those of alkanes?
Assume all compounds have similar molecular weight, chain length, and branching.
Amines tend to have higher boiling points than those of analogous alkanes.
This trend exists because amines can hydrogen bond, while alkanes cannot. Hydrogen bonding is a strong intermolecular force, and stronger intermolecular forces increase boiling and melting points.
What type of reaction is an amide formation?
An amide formation is a nucleophilic substitution reaction in which an amine attacks a carbonyl carbon and displaces a good leaving group.

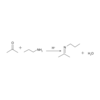
As shown below, an amine can attack an acyl chloride and displace chlorine, forming an amide.
What product will be formed in this reaction?

This reaction will form an amide.

The reaction shown here is a nucleophilic substitution in which the secondary amine attacks the carbonyl carbon. Chloride, a good leaving group, is displaced.
What type of group is added in the acylation of an amine?
In an amine acylation, an acyl group (RCO) is added to an amine, creating an amide.
This reaction is similar to regular amide formation.
What product will be formed in this reaction?

This reaction will form an amide.

This reaction is an acylation, in which the amine acts as a nucleophile and attacks the acyl chloride. Chloride ion, a good leaving group, is displaced.
What type of group is added in the alkylation of an amine?
In an amine alkylation, an alkyl group (R) is added to an amine.
Typically, this reaction will involve nucleophilic substitution, in which an amine attacks an alkyl halide. Alkylations can occur multiple times.
How does the alkylation of an amine affect the nucleophilicity of the final product?
Alkylation increases the nucleophilicity of the resulting amine.
This trend occurs because alkyl groups are electron-donating, and the greater the electron density on the nitrogen atom, the more likely it will be to attack electropositive groups.
Why do exhaustive alkylations of amines by alkyl halides readily occur?
Since alkylation increases the nucleophilicity of the amine, each subsequent reaction becomes increasingly likely to occur.
The amine will continue to perform nucleophilic attacks on the alkyl halides until the amine is fully alkylated.
What product(s) can potentially form in this reaction?

This reaction can form triethylamine, diethylamine, and ethylamine.

Multiple products can form because multiple alkylations occur. Alkylation increases the nucleophilicity of the amine, resulting in further alkylations whenever possible.
Briefly describe what occurs in a Hofmann elimination.
In a Hofmann elimination, a non-terminal amine is exhaustively methylated. The methylated amine acts as a leaving group in an elimination reaction, forming a double bond.
How do Hofmann and Zaitsev eliminations differ?
In a Hofmann elimination, the less substituted double bond is formed.
In a Zaitsev elimination, the more substituted double bond is formed.
What product will be formed in this reaction?

This reaction will form propene (and trimethylamine).

The reaction shown is a Hofmann elimination. In the first step, the amine is exhaustively methylated. It is then eliminated, creating a double bond, in the second step.
What, if any, acid/base properties does an amine have?
Amines are basic, meaning that they readily gain protons if possible.
Ammonia, NH3, is a weak base that commonly appears on the MCAT. NH2- is also a base, though it is much stronger.
What product will form when an amine is reacted with a weak base?
Nothing will occur.
Amines are basic and are not readily deprotonated, so they are unlikely to react with a weak base. However, since they are easily protonated, they would react with an acid.
What properties of an amine’s substituents can affect its basicity?
Properties that affect basicity include:
- The electron-donating or withdrawing nature of substituents on the amine
- The solvability of the amine in its protonated form
- The steric hindrance of the substituents on the amine
How does an alkyl substituent affect the basicity of an amine?
Alkyl substituents increase the basicity of amines because alkyl groups are electron donors.
Electron-donating groups increase an amine’s electron density, making it more prone to accept a proton. On the other hand, electron-withdrawing groups decrease basicity. The opposite of this trend is true for acidity.