Biological Molecules Flashcards
With the biological focus of the new MCAT, having an understanding of carbohydrates, lipids, and amino acids has become especially important. Use these cards to familiarize yourself with the difference between alpha and beta anomers, reducing sugars, and much more. (44 cards)
What features define a carbohydrate?
A carbohydrate is an organic compound with a backbone of at least 3 carbons, typically with a C:H:O ratio of 1:2:1. Carbohydrates consist of either aldehydes or ketones with other carbons in the chain bound to hydroxyl groups.
Exceptions to the 1:2:1 ratio exist for “deoxy” sugars, in which a hydroxyl group has been replaced by a hydrogen.
Carbohydrate structure can be shown in many ways. What name is given to the method of depiction below?

This is a Fischer projection. Straight-chain carbohydrates are often shown in this form.
On any Fischer projection, horizontal lines are considered to point out of the page (“wedges”), while vertical lines point into the page (“dashes”). Thus, this method can accurately convey stereochemistry.
Carbohydrate structure can be shown in many ways. What name is given to the method of depiction below?

This is a Haworth projection. Carbohydrates in the ring form are often shown in this manner.
The relative positions of substituents as “cis” or “trans” can be determined by whether they point upward or downward from the face of the ring.
What is the difference between an aldose and a ketose?
Aldoses are sugar molecules that contain a single aldehyde group, while ketoses contain a single ketone.
Note that both aldoses and ketoses are monosaccharides.
Briefly describe the steps for naming a straight-chain carbohydrate.
- The compound is first classified as either an aldose or a ketose.
- Next, the number of carbons is counted (di, tri, quatr, pent) and included after the aldo- or keto- prefix.
- Finally, the -ose suffix, used to denote a carbohydrate, is attached to the name.
Note that there should be no spaces between the prefix, numerical identifier, and suffix.
According to simple carbohydrate nomenclature, what general name can be given to this molecule?

This molecule is an aldopentose.
This carbohydrate has an aldehyde group, which should be denoted by the prefix “aldo-.” As a five-carbon sugar, it should be named using the Greek term “pent.” Finally, the “-ose” suffix identifies the molecule as a carbohydrate.
How are cyclic carbohydrates named?
Cyclic carbohydrates are named based on the number of members in the ring. The two main categories of cyclic carbohydrates are the five-membered furanoses and the six-membered pyranoses.
To denote a carbohydrate in the ring form, remove “-se” from the carbohydrate name and replace it with the suffix “-furanose” or “-pyranose,” as is appropriate.
With regard to its cyclic structure, what general name can be given to this molecule?
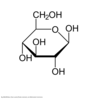
This molecule is a pyranose.
The carbohydrate has a six-membered ring form, so it is a pyranose. Five-membered rings are known as furanoses.
What is the common name of this molecule?

This molecule is glucose.
Glucose, a hexose, is the most common carbohydrate tested on the MCAT. Its Fischer projection can be identified by the orientation of its hydroxyl groups. From top to bottom, the -OH groups will be oriented either “right, left, right, right” (D-glucose) or “left, right, left, left” (L-glucose).
What is the common name of this molecule?

This molecule is fructose.
Fructose, a pentose, is often mentioned on the MCAT. Its Fischer projection can be identified by the orientation of its hydroxyl groups. From top to bottom, the -OH groups will be oriented either “left, right, right” (D-fructose) or “right, left, left” (L-fructose).
What are the three main structural ways in which carbohydrates are classified?
- As aldoses (aldehyde-containing carbohydrates) or ketoses (ketone-containing carbohydrates)
- As furanoses (five-membered rings) or pyranoses (six-membered rings)
- As regular sugars (those with oxygen atoms bound to each carbon) or deoxy sugars (those missing one oxygen atom)
What is the difference between a monosaccharide and a disaccharide?
A monosaccharide is composed of one cyclic sugar subunit, while a disaccharide is composed of two cyclic sugars connected by a glycosidic linkage.
A polysaccharide, then, consists of more than two sugar subunits bound together.
Name three common monosaccharides.
Three of the most common monosaccharides to appear on the MCAT are glucose, fructose, and galactose. Ribose and deoxyribose are also monosaccharides.
Monosaccharides can be connected by glycosidic linkages to form longer sugar chains.
Name three common disaccharides.
Three of the most common disaccharides to appear on the MCAT are sucrose, lactose, and maltose.
In general, disaccharides consist of two sugar subunits bound by a glycosidic linkage.
Which sugar monomers comprise sucrose, lactose, and maltose, respectively?
- Sucrose is composed of fructose and glucose
- Lactose is composed of glucose and galactose
- Maltose is composed of two glucose monomers

Which carbon is used to determine the absolute configuration of a carbohydrate?
The absolute configuration of a straight-chain carbohydrate is determined by the chirality of the carbon stereocenter that is farthest from the carbonyl carbon.
In other words, the absolute configuration depends on the highest-numbered chiral carbon in the molecule.
What determines whether the absolute configuration of a carbohydrate is D or L?
Any carbohydrate depicted as a Fischer projection:
- is classified as a D sugar if the -OH on the highest-numbered chiral carbon is pointing to the right.
- is classified as an L sugar if that same -OH is instead pointing to the left.
What is the absolute configuration of this carbohydrate?

This carbohydrate has a D configuration.

Look at the -OH on the chiral carbon farthest from the carbonyl group. Since that -OH is pointing to the right on the Fischer projection, this must be a D sugar.
What are epimers?
Epimers are two molecules that differ in configuration at a single chiral carbon.
Epimers are a special type of diastereomer. They are especially important in relation to sugars.
What type(s) of isomer are the carbohydrates below?
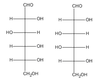
These carbohydrates are epimers, since they differ in configuration at only one position: the third carbon from the carbonyl.
Like all epimers, these molecules are also diastereomers.
Describe the stereochemical relationship between carbohydrates 1, 2, and 3.

- 1 and 2 are epimers differing at the chiral carbon closest to the carbonyl. Like all epimers, they are also diastereomers.
- 2 and 3 are enantiomers, or nonsuperimposable mirror images of each other. They differ in configuration at all chiral centers.
- 1 and 3 are diastereomers, since they differ in configuration at 3 of 4 chiral carbons. Diastereomers are stereoisomers which differ at some (but not all) stereocenters.
Describe the stereochemical relationship between carbohydrates 1, 2, and 3.

- 1 and 2 are epimers differing at the chiral carbon closest to the carbonyl. Like all epimers, 1 and 2 are also diastereomers.
- 1 and 3 are epimers differing at the chiral carbon second from the carbonyl. Again, 1 and 3 are also diastereomers.
- 2 and 3 have no formal relationship.
Two carbohydrates are structurally identical but differ in configuration at three of their four chiral centers. What type(s) of isomer are these molecules?
These molecules are diastereomers, a broad term for stereoisomers that are not enantiomers.
Specifically, these molecules cannot be enantiomers since they do not have opposite configurations at all of their chiral carbons. They also are not epimers, which only differ in configuration at one position.
For a carbohydrate to isomerize between the straight-chain and ring forms, what reaction must take place?
In the straight-chain form, the -OH on the chiral carbon farthest from the carbonyl acts as a nucleophile and attacks the carbonyl carbon. This forms a cyclic structure.
In the proccess, the attacking hydroxyl loses its proton and the carbonyl oxygen becomes protonated.












