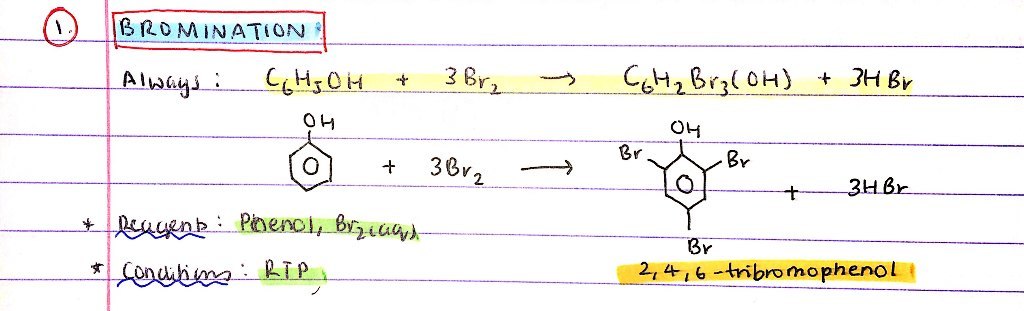What is a aromatic compound?
Contains benzene
Formula of benzene = ?
- Look and smell?
- Where is it found?
- What is it an example of?
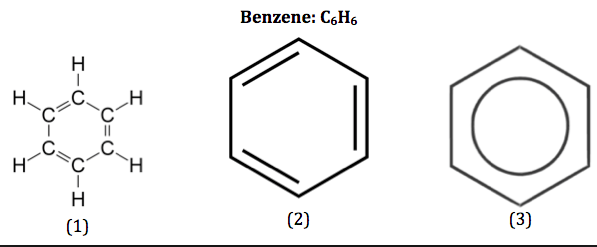
Benzene = C6H6
- Colourless, sweet smelling and highly flammable liquid
- Found:
- Naturally in crude oil - is a component of petrol
- In cigarette smoke
- It’s a carcinogen = capable of causing cancer
What is a carcinogen?
Something that has the ability to cause cancer
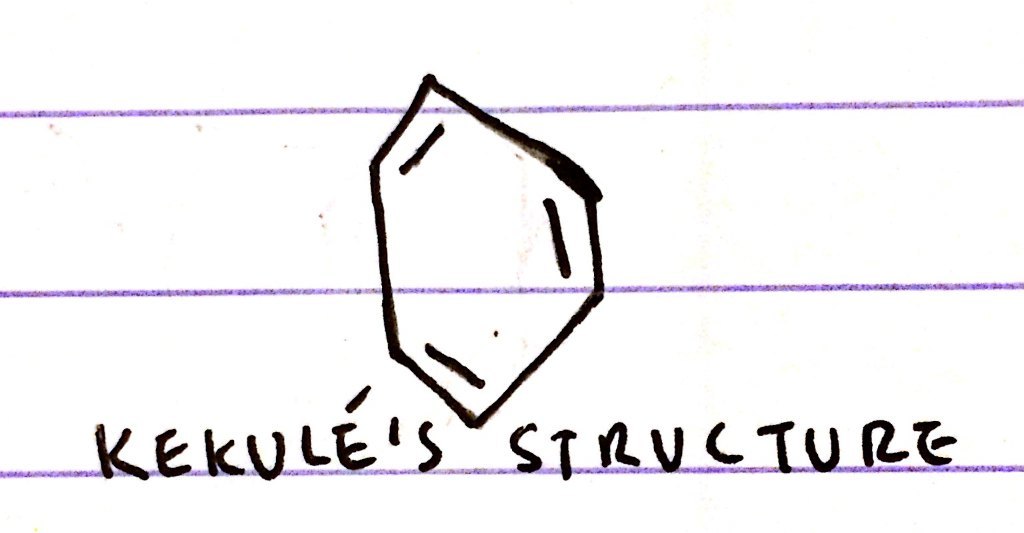
What did kekulé think benzene looked like?
- 6C’s and 6H’s arranged in a ring - C6H6
- 3 x C-C and 3 x C=C
- C-C bonds are slightly longer than C=C bonds
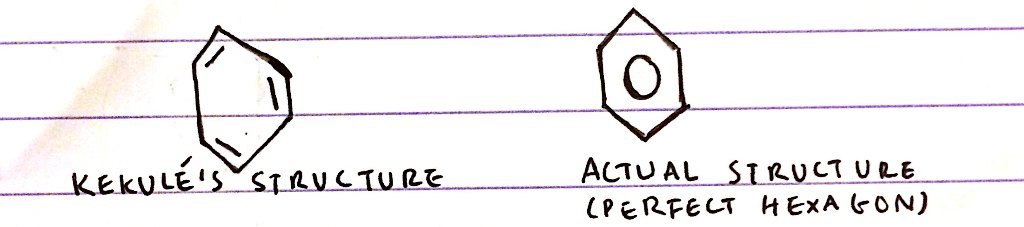
3 pieces of evidence against kekulé’s structure of benzene
- Benzene has equal bond lengths forming a regular hexagonal structure
- Evidence from x-ray crystallography and tunnelling electron microscopy
- Kekulé’s struture would’ve lead to the compound being skewered
- Benzene doesn’t undergo electrophilic addition reactions with bromine
- Doesn’t decolourise with bromine water
- Benzene has a less exotheric enthalpy change of hydrogenation than expected
4.
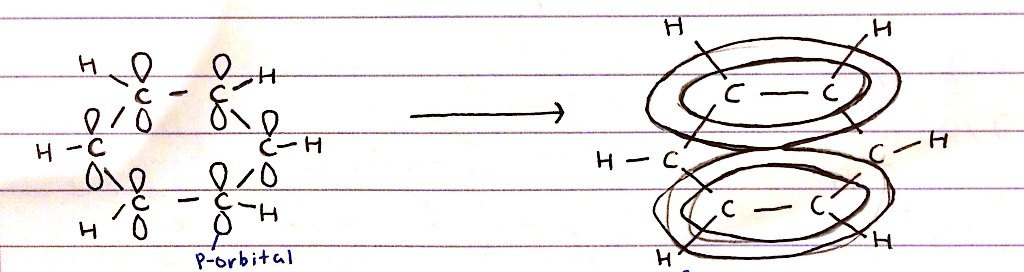
How do the delocalised rings of electrons form in benzene?
- P-orbitals are present above and below the plane of the carbon ring
- Sideways overlap of p-orbitals create a delocalised rings of electrons above and below the plane of the benzene ring
- ∴ creates a system of Π bonds spread over the 6Cs in the ring
3 pieces of evidence to support delocalised structure of benzene
Bond lengths are all the same:
- Kekulé’s structure would lead to 3 long + 3 short bonds ∴ it would be skewered
- In reality bond lengths are half way between C-C and C=C
Less exothermic enthalpy change of hydrogenation than expected:
- Need more energy to break bonds ∴ benzene is more stable than expected
Doesn’t undergo electrophilic addition reactions with Br(aq):
- Kekulé expected it to delcolourise Br(aq) but it didn’t
- If it did react with benzene it would lose the ring structure
- Undergoes electrophilic substitution reactions instead
Common question!

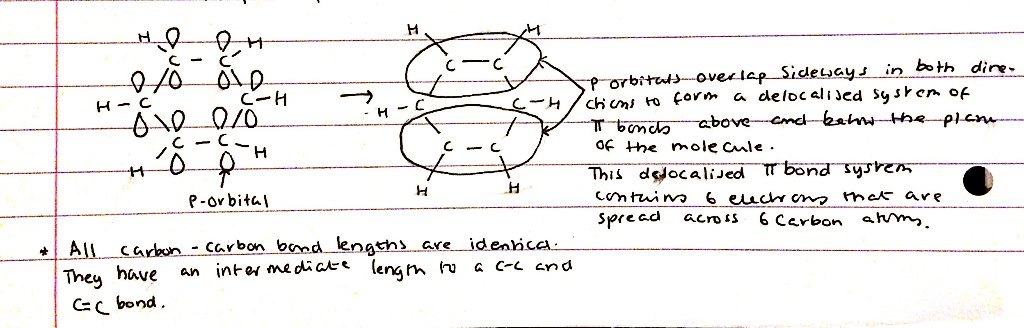
- Benzene has a formula C6H6. It’s a planar and is a regular hexagon
- Main framework of molecules is helf together by 6 C-H σ bonds and 6 C-H σ bonds, each containing 2 electrons
- This leaves 1 outer electron on each carbon atom in a p-orbital orientated perpendicular to the plane of the molecule
- DIAGRAM:
- P-orbitals overlap sideways in both directions to form a delocalised system of π bonds above and below the plane of the molecule
- This delocalised π system contains 6 electrons that are spread across 6 C atoms
- All carbon - carbon bonds are equal in length. They’re an intermediate length to a C-C and a C=C bond
What type of reactions do benzene undergo?
Electrophilic substitution
What are the 4 types of reactions that you need to know for benzene?
- Nitration
- Halogenation
- Alkylation
- Acylation
Nitration of benzene:
- Reagents
- Conditions
- Mechanism
- R = Benzene, conc HNO3(aq), conc H2SO4(aq) catalyst
- C = < 50ºC (for mono substitution) > 50ºC gives a di substitution
Mechanism:
- Form NO2+ electrophile:
- HNO3(aq) + H2SO4(aq) → NO2+(aq) + HSO4-(aq) + H2O(l)
- Reaction mechanism - diagram
- Reform catalyst:
- H+(aq) + HSO4-(aq) → H2SO4(aq)
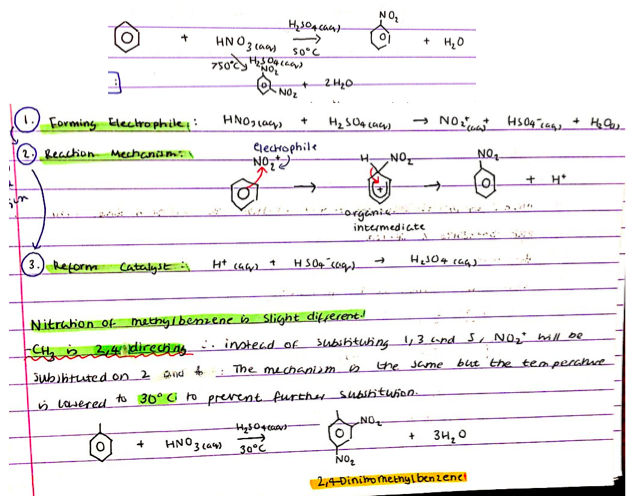
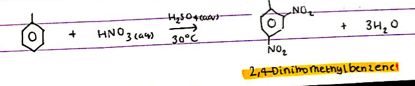
What is different about nitration of methylbenzene to nitration of benzene?
What is different about the conditions?
CH3 is 2,4 directing ∴ instead of substituting NO2+ on carbon 1, 3 and 5 it is substituted onto carbons 2 + 4.
The reagents remain the same as nitration fo benzene:
- Benzene, conc HNO3(aq), conc H2SO4(aq) catalyst
The temperature is lowered to 30ºC
What is TNT?
An explosive - it’s an abbreviation of trinitroluene
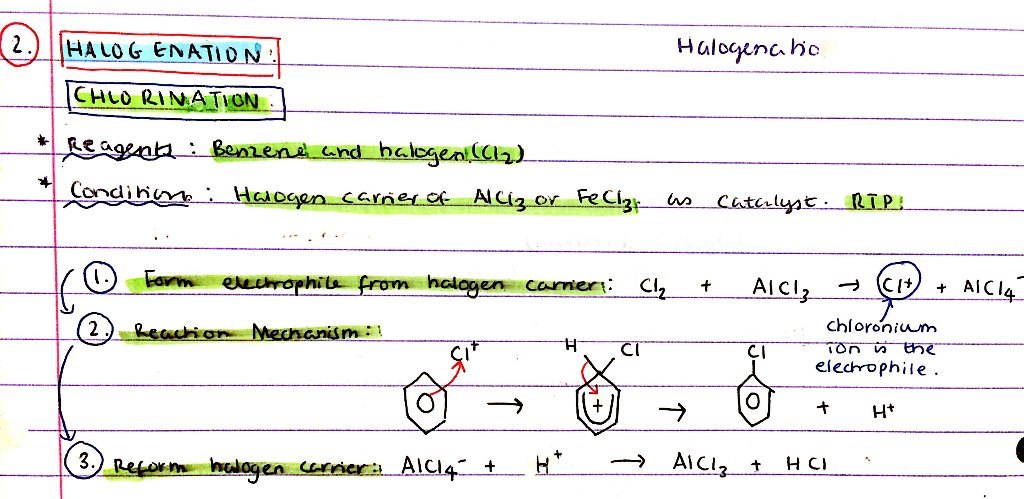
Halogenation of benzene - chlorination:
- Reagents
- Conditions
- Mechanism
- R = Benzene, Cl2
- C = Halogen carrier of AlCl3 or FeCl3 as a catalyst, RTP
Mechanism:
- Form Cl+ electrophile from halogen carrier:
- Cl2 + AlCl3 → Cl+ + AlCl4-
- Reaction mechanism - diagram
- Reform halogen carrier:
- AlCl4- + H+ → AlCl3 + HCl
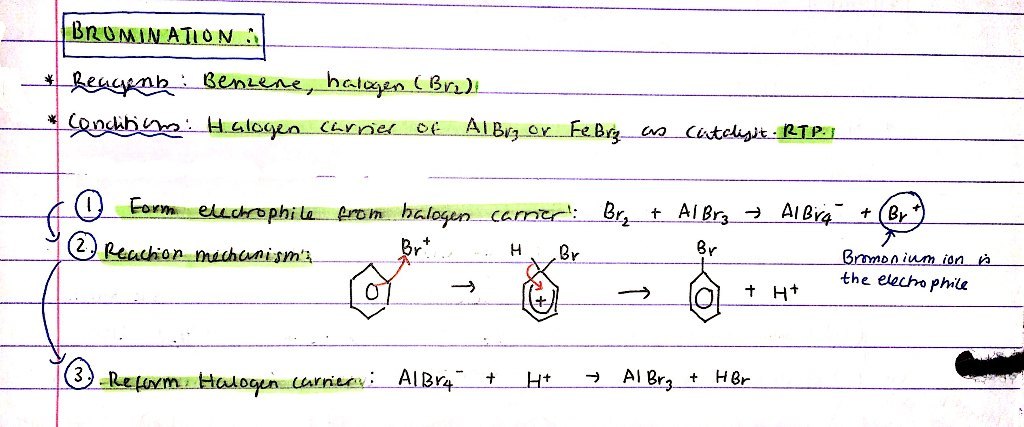
Halogenation of benzene - bromination:
- Reagents
- Conditions
- Mechanism
- R = Benzene, Cl2
- C = Halogen carrier of AlCl3 or FeCl3 as a catalyst, RTP
Mechanism:
- Form Br+ electrophile from halogen carrier:
- Br2 + AlCl3 → Br+ + AlCl4-
- Reaction mechanism - diagram
- Reform halogen carrier:
- AlCl4- + H+ → AlCl3 + HCl
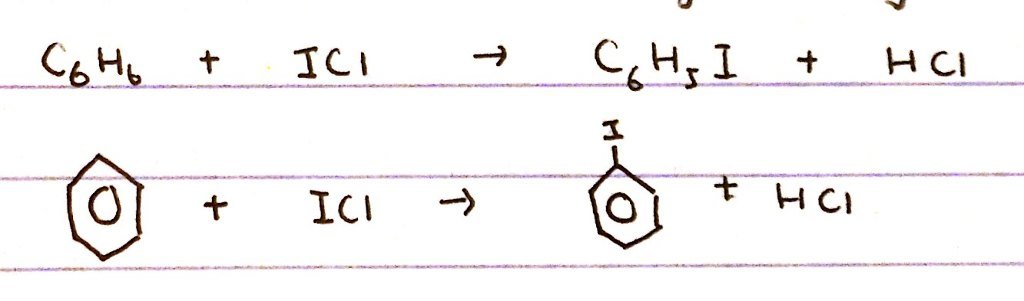
Why does I2 not substitute on benzene like Br2 and Cl2 does?
How can iodobenzene be made?
- I2 is too unreactive to sunstitue on benzene - even in the presence of a catalyst
- BUT iodobenze can be made by reacting benzene + iodochloride
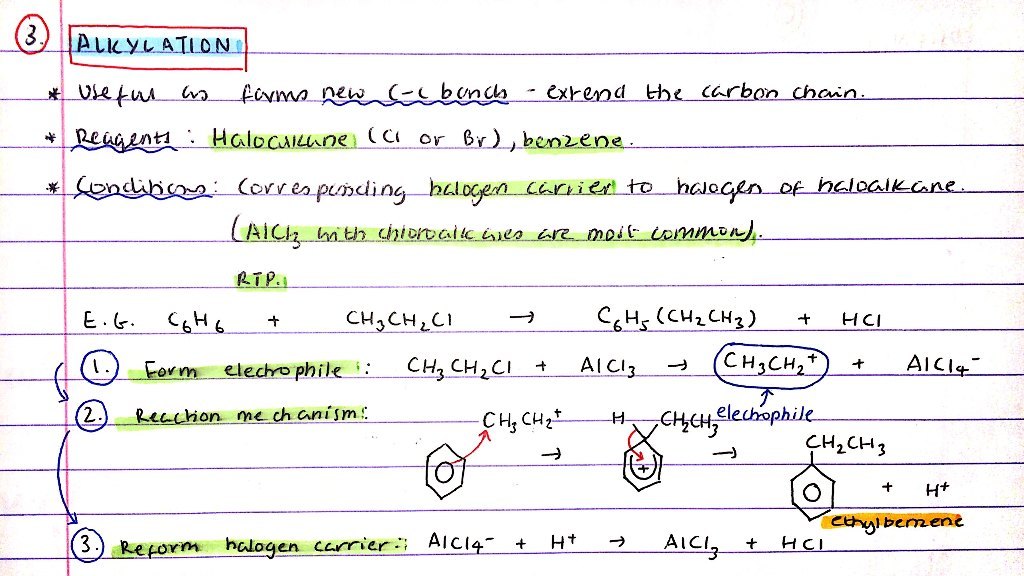
Alkylation of benzene:
- What is this reaction useful for?
- Reagents
- Conditions
- Mechanism
- Useful as forms new C-C bonds to extend the carbon chain
- R = Haloalkane (Cl or Br), benzene
- C = Corresponding halogen carrier - AlCl3 / Chloroalkanes are most common
Mechanism:
- Form Cl+ electrophile from halogen carrier:
- Cl2 + AlCl3 → Cl+ + AlCl4-
- Reaction mechanism - diagram
- Reform halogen carrier:
- AlCl4- + H+ → AlCl3 + HCl
Acylation of benzene
- What does it form?
- Reagents
- Conditions
- Mechanism
- Forms aromatic ketones by substituting a acyl chloride onto benzene
- R = Acyl chloride, benzene
- C = AlCl3 halogen carrier as catalyst, RTP
Mechanism:
- Form Cl+ electrophile from halogen carrier:
- Cl2 + AlCl3 → Cl+ + AlCl4-
- Reaction mechanism - diagram
- Reform halogen carrier:
- AlCl4- + H+ → AlCl3 + HCl
Define Electrophile
Electrophile = An atom or group of atoms which is attracted to an electron rich centre or atom, where it accepts a pair of electrons to form a covalent bond
Phenol:
- Appearence at RTP
- Reaction with water - solubility in water?
- Add NaOH(aq) to phenol in water
- Add Na2CO3(s) to phenol inw ater
- Add bromine water to solid phenol
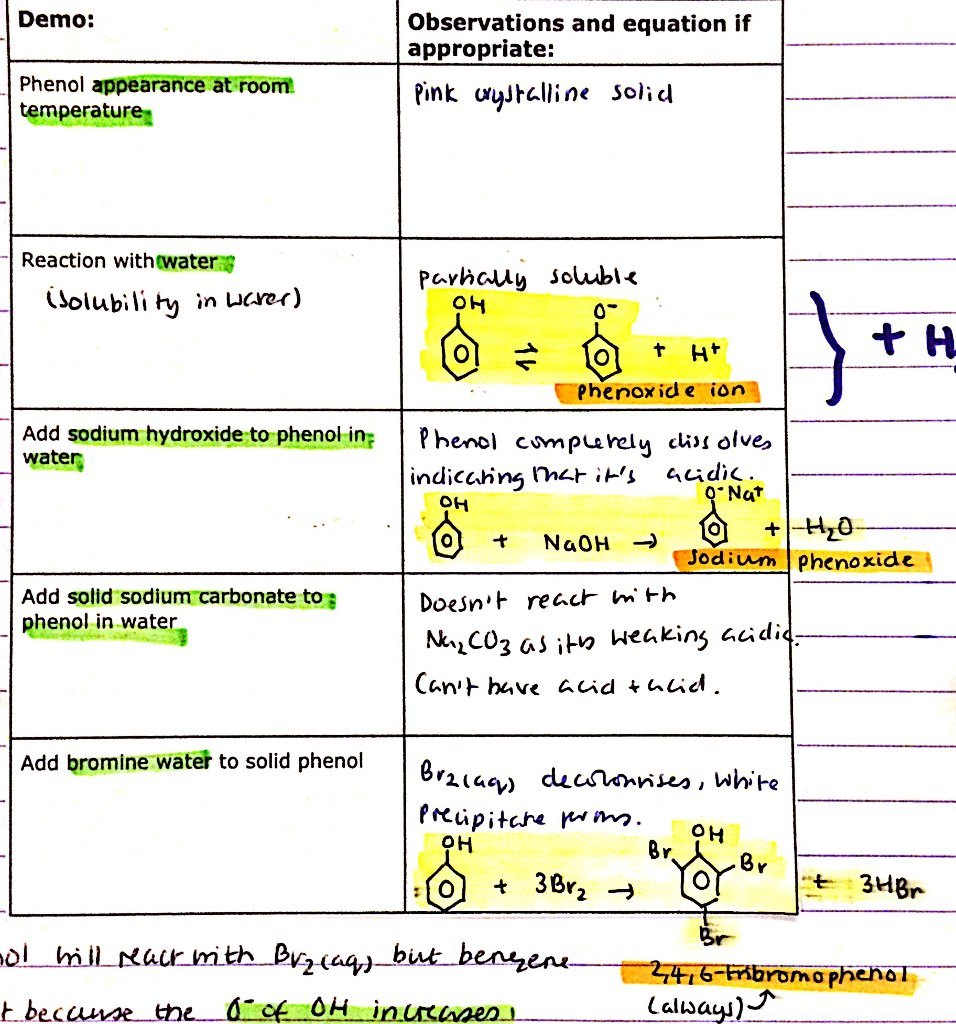
Why does phenol react with bromine water but benzene doesn’t?
O- of the OH- increases negative charge when it mixes of of it’s electrons with the electron density, increased negative charge attracts Br2 more
What type of reactions do phenol undergo?
Electrophilic substitution reactions
Name 2 types of electrophilic substitution reactions you need to know for phenol
- Nitration
- Bromination
Bromination of phenol:
- Reagents
- Conditions
- Products
- R = Phenol, Br2(aq)
- C = RTP
Always forms 2,4,6 - tribromophenol